 
Antoszczyszyn and Michalska reported the potential risk of environmental contamination by mercury contained in Polish coal mining waste. studied the mercury exposure and effects across the life span. reviewed about the mercury speciation, transformation, and transportation in soils, atmospheric flux, and implications for risk management. 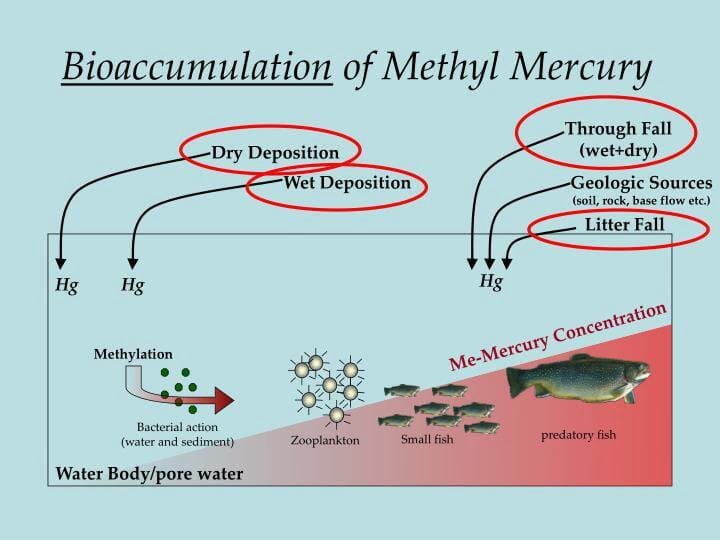
reviewed the sources, pathways, and effects of mercury in global environment. Bernhoft reviewed about the mercury toxicity and its treatments. reviewed the mercury exposure and health effects of dental personnel. Methylmercury is the best example it mixes with the food chain. Organic mercury: It is in the forms of aryl and alkyl. This is present in crystal form, and it is used in pesticides and antiseptics. Inorganic mercury: It occurs in several forms: metallic (Hg), mercurous (Hg 1+), or mercuric form (Hg 2+). It is used in thermometers and fluorescent bulbs. The chemical forms of mercury in the environment are broadly divided into three, i.e., (1) elemental mercury, (2) inorganic salts, and (3) organic compounds.Įlemental mercury: The atmosphere consists of mercury in elemental form, and at room temperature it is present in liquid form. Mercury in the environment can exist in different forms, such as mercurous ion (Hg +1), mercuric ion (Hg +2), methylmercury (CH 3Hg +), ethylmercury (C 2H 5Hg +), and phenylmercury (C 6H 5Hg +). Mercury is a metal that appears as silver balls, and its liquid form is called elemental mercury. A recent review reported about the worldwide and regional time trends in total mercury levels in the human blood and breast milk and their associations with health effects. The Minamata Convention on Mercury entered into force from 2017, regarding the protection of human and environmental health. Mercury is a pollutant of global concern. According to the US Environmental Protection Agency, the safe limit of mercury ion in drinking water is 10 nM to avoid the serious health problems to humans. Mercury (Hg) is a naturally occurring element that is toxic in nature. It is very important to measure the concentration of mercury in the environment due to its bioaccumulative property in its elemental and organic forms (i.e., methylmercury) and toxic nature. Mercury is the only metal that exists in liquid state among the total periodic table elements. Mercury has a great affinity with thiol group in proteins and causes many diseases. The absorbed mercury in humans becomes more toxic due to its prolonged half-life and lack of decomposition, and its interaction is not limited with various enzymes and proteins. Once these metals are absorbed into the human body, they can be a threat with more health issues. The metals such as cadmium, mercury, and lead are owing much more interest to environmental scientists due to their accumulation in vital organs of the living beings. Heavy metal contamination is a serious problem to the environment, because they are not only biodegradable but also toxic to living organisms. The present chapter describes the mercury sources, types of exposures, types of poisoning, treatments, and preventive measures of mercury poisoning. Mercury poisoning effects on the human body are not limited to reddishness of hands and feet renal failures cardiovascular, liver, brain, and hormonal issues and intestinal ulceration. Symptoms of mercury poisoning depend on the type, dose, method, and duration of exposure. 
Mercury poisoning occurs by exposure to mercury, i.e., acute and chronic exposures. Mercury is the only metal that exists in liquid state at normal temperature and pressure. Mercury in +2 state is more poisonous due to high solubility. But the liquid mercury can vaporize, and gaseous mercury becomes poisonous due to its nature of being absorbed into the blood. Liquid metal state mercury (Hg) has little to no solubility and is not poisonous. It is also called as quicksilver due to its fast movement and silvery tinge. Mercury was the name of the Roman messenger of god who can move really fast. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
